|
As a consequence, confusion existed regarding atomic weights and molecular formulas for about half a century.Īs early as 1805, Gay-Lussac and von Humboldt showed that water is formed of two volumes of hydrogen and one volume of oxygen, and by 1811 Amedeo Avogadro had arrived at the correct interpretation of water's composition, based on what is now called Avogadro's law and the assumption of diatomic elemental molecules. For example, Dalton assumed water's formula to be HO, giving the atomic weight of oxygen as eight times that of hydrogen, instead of the modern value of about 16. John Dalton's original atomic hypothesis assumed that all elements were monatomic and that the atoms in compounds would normally have the simplest atomic ratios with respect to one another. Historical significance ĭiatomic elements played an important role in the elucidation of the concepts of element, atom, and molecule in the 19th century, because some of the most common elements, such as hydrogen, oxygen, and nitrogen, occur as diatomic molecules. Diatomic nitrogen has a triple bond, diatomic oxygen has a double bond, and diatomic hydrogen, fluorine, chlorine, iodine, and bromine all have single bonds. The interstellar medium is dominated by hydrogen atoms.Īll diatomic molecules are linear and characterized by a single parameter which is the bond length or distance between the two atoms. 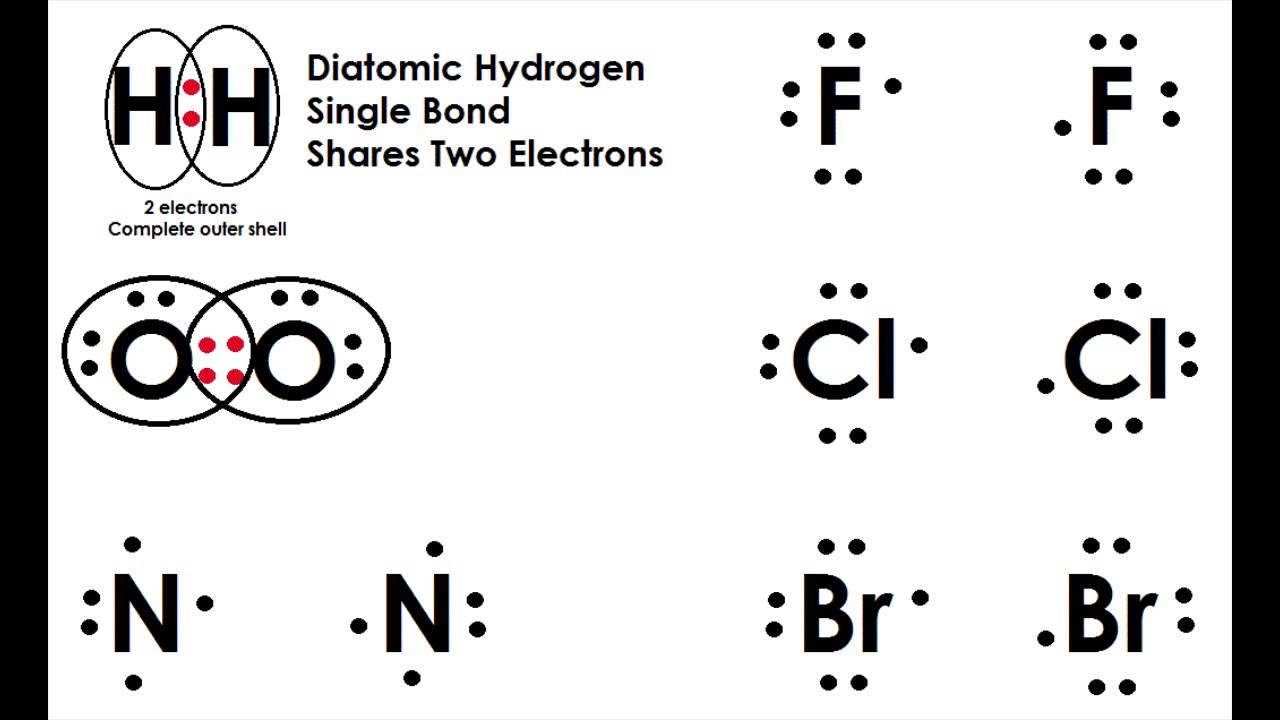
The natural abundance of hydrogen (H 2) in the Earth's atmosphere is only of the order of parts per million, but H 2 is the most abundant diatomic molecule in the universe. About 99% of the Earth's atmosphere is composed of two species of diatomic molecules: nitrogen (78%) and oxygen (21%). Hundreds of diatomic molecules have been identified in the environment of the Earth, in the laboratory, and in interstellar space. Many 1:1 binary compounds are not normally considered diatomic because they are polymeric at room temperature, but they form diatomic molecules when evaporated, for example gaseous MgO, SiO, and many others. Many elements can combine to form heteronuclear diatomic molecules, depending on temperature and pressure.Įxamples are gases carbon monoxide (CO), nitric oxide (NO), and hydrogen chloride (HCl). Dirubidium ( Rb 2) is diatomic.Īll other diatomic molecules are chemical compounds of two different elements. Di tungsten ( W 2) and di molybdenum ( Mo 2) form with sextuple bonds in the gas phase. Dilithium ( Li 2) and disodium ( Na 2) are known in the gas phase. Heating ("cracking") elemental phosphorus gives diphosphorus ( P 2). Other elements form diatomic molecules when evaporated, but these diatomic species repolymerize when cooled. 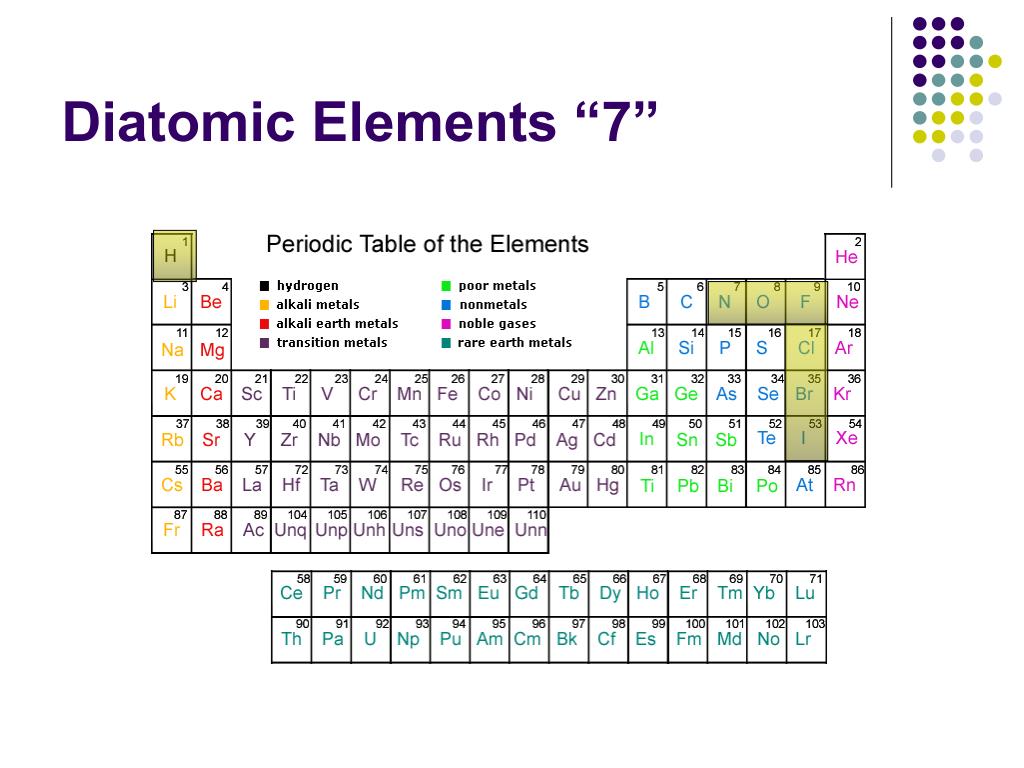
All halogens have been observed as diatomic molecules, except for astatine and tennessine, which are uncertain. Īt slightly elevated temperatures, the halogens bromine ( Br 2) and iodine ( I 2) also form diatomic gases. 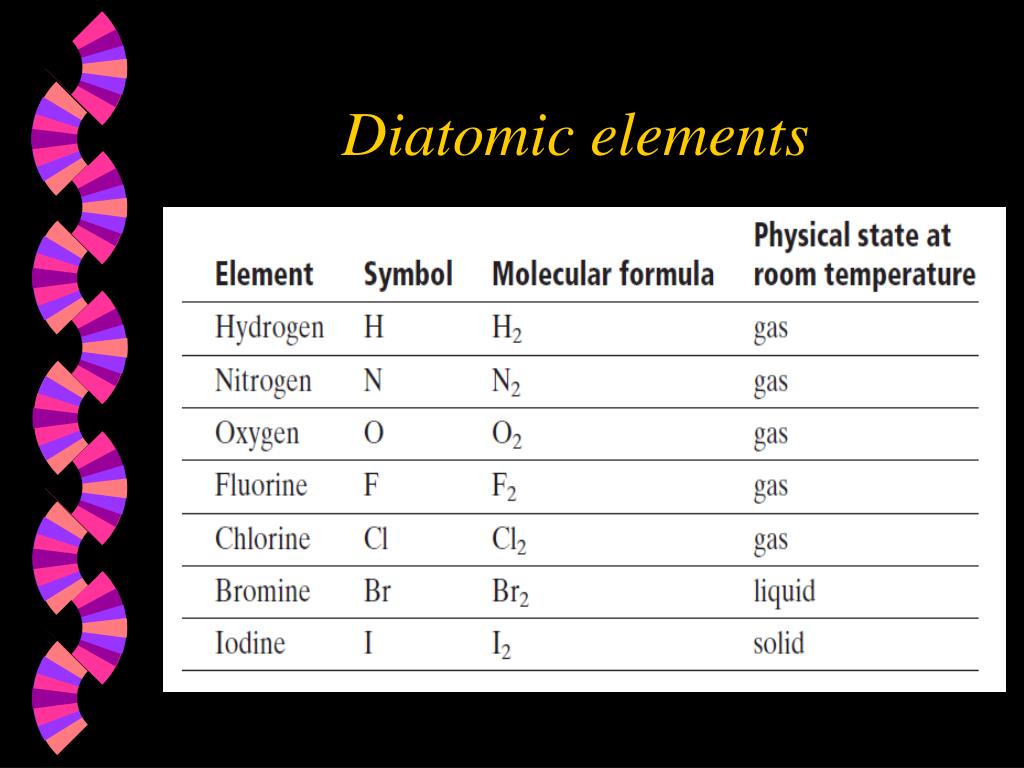
The homonuclear diatomic gases and noble gases together are called "elemental gases" or "molecular gases", to distinguish them from other gases that are chemical compounds. The noble gases ( helium, neon, argon, krypton, xenon, and radon) are also gases at STP, but they are monatomic. 
The only chemical elements that form stable homonuclear diatomic molecules at standard temperature and pressure (STP) (or typical laboratory conditions of 1 bar and 25 ☌) are the gases hydrogen ( H 2), nitrogen ( N 2), oxygen ( O 2), fluorine ( F 2), and chlorine ( Cl 2). A periodic table showing the elements that exist as homonuclear diatomic molecules under typical laboratory conditions.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
